Vinyl sulfonyl fluoride
ethylenesulphonyl fluoride
CAS: 677-25-8
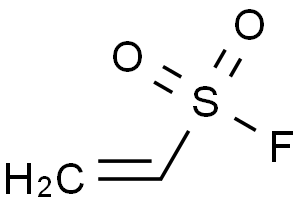
Molecular Formula: C2H3FO2S
Vinyl sulfonyl fluoride - Names and Identifiers
| Name | ethylenesulphonyl fluoride |
| Synonyms | Fluorosulfonylethene Ethenesulfonylfluoride Vinyl sulfonyl fluoride ethylenesulphonyl fluoride Ethenesulfonic acid fluoride |
| CAS | 677-25-8 |
| EINECS | 2116407 |
Vinyl sulfonyl fluoride - Physico-chemical Properties
| Molecular Formula | C2H3FO2S |
| Molar Mass | 110.11 |
| Density | 1.328 g/mL at 25 °C |
| Boling Point | 44-46 °C(Press: 50 Torr) |
| Flash Point | 40℃ |
| Refractive Index | n20/D1.385 |
| Physical and Chemical Properties | EPA Chemical Information Ethenesulfonyl fluoride (677-25-8) |
Vinyl sulfonyl fluoride - Risk and Safety
| Risk Codes | R23/24/25 - Toxic by inhalation, in contact with skin and if swallowed. R34 - Causes burns R68 - Possible risk of irreversible effects |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | UN 2920 3(8) / PGII |
| WGK Germany | 3 |
Vinyl sulfonyl fluoride - Introduction
ethylenesulphonyl fluoride is an organic compound with the chemical formula C2H3FO2S. The following is a description of the nature, use, formulation and safety information of ethylenesulphonyl fluoride:
Nature:
1. Appearance: ethylenesulphonyl fluoride is a colorless liquid.
2. Melting point and boiling point: the melting point of ethylensulphonyl fluoride is about 5 ° C, and the boiling point is about 65 ° C.
3. Density: The density of ethylensulphonyl fluoride is about 1.278g/cm3.
4. Solubility: ethylenesulphonyl fluoride is soluble in most organic solvents, such as methanol, ethanol, acetone and dichloromethane.
Use:
ethylenesulphonyl fluoride is often used as sulfonylation reagent in organic synthesis, and is often used in the synthesis of sulfonyl-containing organic compounds, such as sulfonyl fluoroketone, sulfonimide, etc. It can also be used to prepare sulfonyl fluoride compounds and sulfonic acid esters. In addition, ethylenesulphonyl fluoride can also be used as chemical reagents, organic intermediates and electronic materials.
Method:
The preparation of ethylenesulphonyl fluoride can be obtained by the reaction of fluorinated sulfoxide with hydrogen potassium fluoride or sodium fluoride. The specific preparation method is as follows:
1. Under dry conditions, the fluorinated sulfoxide is reacted with hydrogen potassium fluoride or sodium fluoride.
2. After the reaction is completed, ethylensulphonyl fluoride is obtained by distillation and purification.
Safety Information:
ethylenesulphonyl fluoride has a stimulating effect on the skin, eyes and mucous membranes, and should be rinsed with plenty of water immediately after contact. Wear protective glasses and gloves during use. In addition, ethylenesulphonyl fluoride belongs to toxic chemicals, should avoid inhalation or ingestion, avoid contact with flammable substances. Proper storage and handling of ethylenesulphonyl fluoride is important and safe operating procedures should be followed. Any ethylenesulphonyl fluoride-related operations should be carried out in a well-ventilated environment.
Nature:
1. Appearance: ethylenesulphonyl fluoride is a colorless liquid.
2. Melting point and boiling point: the melting point of ethylensulphonyl fluoride is about 5 ° C, and the boiling point is about 65 ° C.
3. Density: The density of ethylensulphonyl fluoride is about 1.278g/cm3.
4. Solubility: ethylenesulphonyl fluoride is soluble in most organic solvents, such as methanol, ethanol, acetone and dichloromethane.
Use:
ethylenesulphonyl fluoride is often used as sulfonylation reagent in organic synthesis, and is often used in the synthesis of sulfonyl-containing organic compounds, such as sulfonyl fluoroketone, sulfonimide, etc. It can also be used to prepare sulfonyl fluoride compounds and sulfonic acid esters. In addition, ethylenesulphonyl fluoride can also be used as chemical reagents, organic intermediates and electronic materials.
Method:
The preparation of ethylenesulphonyl fluoride can be obtained by the reaction of fluorinated sulfoxide with hydrogen potassium fluoride or sodium fluoride. The specific preparation method is as follows:
1. Under dry conditions, the fluorinated sulfoxide is reacted with hydrogen potassium fluoride or sodium fluoride.
2. After the reaction is completed, ethylensulphonyl fluoride is obtained by distillation and purification.
Safety Information:
ethylenesulphonyl fluoride has a stimulating effect on the skin, eyes and mucous membranes, and should be rinsed with plenty of water immediately after contact. Wear protective glasses and gloves during use. In addition, ethylenesulphonyl fluoride belongs to toxic chemicals, should avoid inhalation or ingestion, avoid contact with flammable substances. Proper storage and handling of ethylenesulphonyl fluoride is important and safe operating procedures should be followed. Any ethylenesulphonyl fluoride-related operations should be carried out in a well-ventilated environment.
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Ethenesulfonyl fluoride 95% Visit Supplier Webpage Request for quotationCAS: 677-25-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Ethylenesulphonyl Fluoride Visit Supplier Webpage Request for quotationCAS: 677-25-8
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: ethylenesulphonyl fluoride Request for quotation
CAS: 677-25-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 677-25-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: ethylenesulphonyl fluoride Request for quotation
CAS: 677-25-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 677-25-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: ethylenesulphonyl fluoride Visit Supplier Webpage Request for quotation
CAS: 677-25-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 677-25-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Ethenesulfonyl fluoride 95% Visit Supplier Webpage Request for quotationCAS: 677-25-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Ethylenesulphonyl Fluoride Visit Supplier Webpage Request for quotationCAS: 677-25-8
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: ethylenesulphonyl fluoride Request for quotation
CAS: 677-25-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 677-25-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: ethylenesulphonyl fluoride Request for quotation
CAS: 677-25-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 677-25-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: ethylenesulphonyl fluoride Visit Supplier Webpage Request for quotation
CAS: 677-25-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 677-25-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




